
Kill COVID-19 on Contact
The Environmental Protection Agency approved PDI's Sani-Prime Germicidal Spray for use against the coronavirus. The spray deactivates SARS-CoV-2 — the virus that causes COVID-19 — after one minute of contact time on hard, nonporous surfaces, according to the new labeling. A combination of quaternary ammonium, isopropyl alcohol (IPA) and ethanol provides fast-acting disinfecting action against bacteria, fungus and virucidal agents, as well as multidrug-resistant organisms such as MRSA, CRE and VRE. The 32-ounce bottle features an easy-to-use trigger spray.
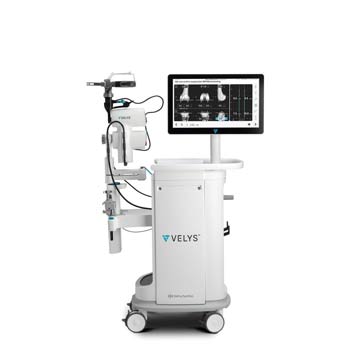
Simplify Total Knee Replacements
The VELYS Robotic-Assisted Solution from DePuy Synthes received 510(k) FDA clearance last month. The table-mounted platform helps surgeons plan, execute and perform total knee replacements by allowing for more accurate bone cuts and improved alignment of implants. It also provides surgeons with gap balance data to help them visualize and predict post-op joint stability. The platform is designed for use with the company’s ATTUNE Total Knee System, which improves the joint’s stability and motion by working in concert with a patient’s specific anatomy.

Robotic Option for GYN Surgeons
The Hominis Surgical System from Memic Innovative Surgery became the first FDA-approved device for transvaginal robotic hysterectomy, an approach associated with fewer complications and shorter surgery and recovery times. Surgeons benefit from a combination of ergonomic controls and advanced surgical capabilities provided by miniature robotic arms that mimic human dexterity for articulation, rotation and maneuverability. With the arms fully retroflexed, the uterus is accessed from the top, a technique familiar to gynecological surgeons.
.svg?sfvrsn=be606e78_3)
.svg?sfvrsn=56b2f850_5)